
Chemistry, 07.09.2021 19:40 jpatte2oye8qv
The polyatomic nitrate ion (NO3−) is present in both the products and the reactants of the chemical equation shown below.
Ba(NO3)2 + 2LiOH → Ba(OH)2 + 2LiNO3
How many nitrate ions are in the products and the reactants of the chemical equation?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

Chemistry, 22.06.2019 18:30
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1
You know the right answer?
The polyatomic nitrate ion (NO3−) is present in both the products and the reactants of the chemical...
Questions


Mathematics, 02.08.2019 14:00





Mathematics, 02.08.2019 14:00


History, 02.08.2019 14:00


Mathematics, 02.08.2019 14:00


Mathematics, 02.08.2019 14:00

Mathematics, 02.08.2019 14:00






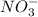 , so we just need to count how many of them are on each side of the equation.
, so we just need to count how many of them are on each side of the equation.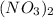 , which shows that there are two nitrate ions in the reactant side (as seen by the little 2).
, which shows that there are two nitrate ions in the reactant side (as seen by the little 2).

