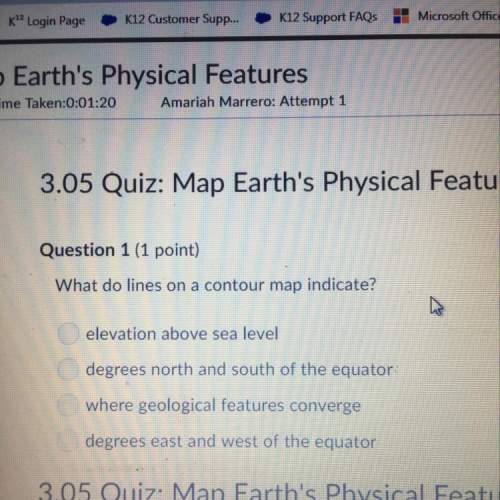
Chemistry, 08.09.2021 19:50 hervinlouis014
Compounds containing potassium and oxygen
are compared. Analysis shows that for each
1.00 g of O, the compounds have 1.22 g, 2.44 g,
and 4.89 g of K, respectively. Show how these
data support the law of multiple proportions.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Temperature and kinetic energy are proportional. a) adirectly b) directly c) indirectly
Answers: 2


Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1
You know the right answer?
Compounds containing potassium and oxygen
are compared. Analysis shows that for each
1.00 g...
1.00 g...
Questions



Mathematics, 26.05.2020 02:03

Mathematics, 26.05.2020 02:03



Mathematics, 26.05.2020 02:03











Arts, 26.05.2020 02:03

Mathematics, 26.05.2020 02:03

Social Studies, 26.05.2020 02:03