Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

You know the right answer?
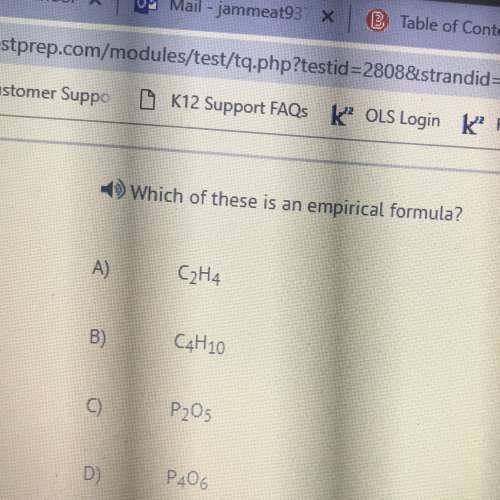
41. Which one of the relationship is wrong? 1) 2 Vapour Density = Mol. Mass 2) At. Mass = Eq. mass x...
Questions

History, 08.12.2019 21:31

Mathematics, 08.12.2019 21:31



Mathematics, 08.12.2019 21:31


Mathematics, 08.12.2019 21:31


History, 08.12.2019 21:31



Chemistry, 08.12.2019 21:31

Mathematics, 08.12.2019 21:31


Physics, 08.12.2019 21:31


Geography, 08.12.2019 21:31



History, 08.12.2019 21:31