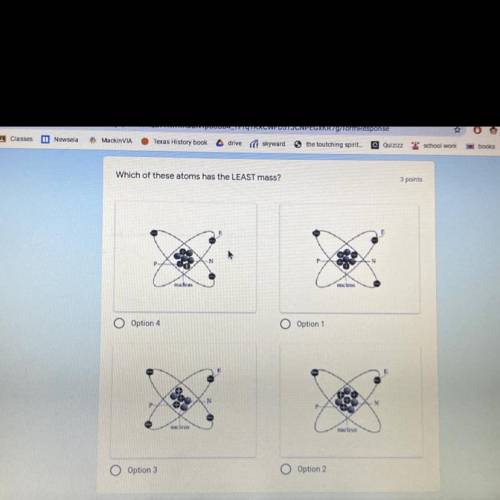
Which of these atoms has less mass
...

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1


Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2
You know the right answer?
Questions

History, 11.03.2021 01:00

Mathematics, 11.03.2021 01:00

Mathematics, 11.03.2021 01:00

Mathematics, 11.03.2021 01:00




Mathematics, 11.03.2021 01:00

Mathematics, 11.03.2021 01:00

World Languages, 11.03.2021 01:00


SAT, 11.03.2021 01:00


Mathematics, 11.03.2021 01:00



Mathematics, 11.03.2021 01:00



Mathematics, 11.03.2021 01:00