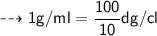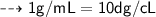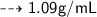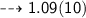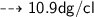Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 22.06.2019 19:50
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2

Chemistry, 23.06.2019 00:00
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3
You know the right answer?
Convert 1.09 g/mL to dg/cL...
Questions

Mathematics, 14.01.2020 12:31


English, 14.01.2020 12:31


History, 14.01.2020 12:31

Chemistry, 14.01.2020 12:31






Physics, 14.01.2020 12:31



History, 14.01.2020 12:31





Mathematics, 14.01.2020 12:31