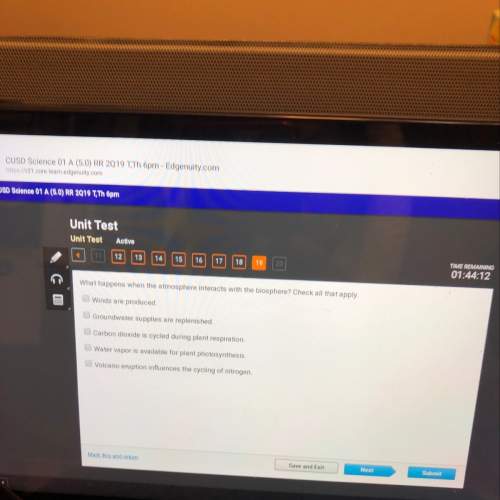
Chemistry, 05.10.2021 18:20 pencil8184
hydrofluoric acid + sodium hydroxide → sodium fluoride + waterHF+NaOH → NaF+H2OWhat are the reactants and products in this equation?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
1. calculate the approximate enthalpy of the reaction in joules. estimate that 1.0 ml of vinegar has the same thermal mass as 1.0 ml of water. iqnore the thermal mass of th sodium bicarbonate. note: it takes about 4.2 joules () to change 1.0 gram (1.0ml) of water 1.0 c
Answers: 2

Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3

Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1
You know the right answer?
hydrofluoric acid + sodium hydroxide → sodium fluoride + waterHF+NaOH → NaF+H2OWhat are the reactant...
Questions


Mathematics, 22.10.2020 01:01

Mathematics, 22.10.2020 01:01

Physics, 22.10.2020 01:01

Mathematics, 22.10.2020 01:01

Geography, 22.10.2020 01:01

Mathematics, 22.10.2020 01:01


Geography, 22.10.2020 01:01


History, 22.10.2020 01:01

English, 22.10.2020 01:01


Mathematics, 22.10.2020 01:01

Mathematics, 22.10.2020 01:01


Mathematics, 22.10.2020 01:01


Health, 22.10.2020 01:01