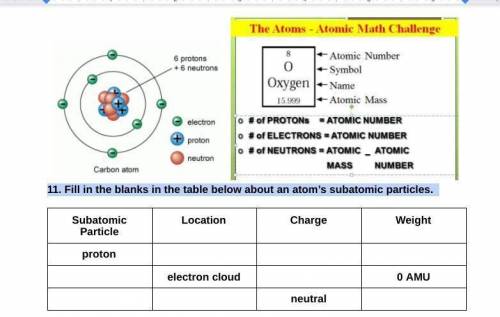
11. Fill in the blanks in the table below about an atom’s subatomic particles.
...


Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1


You know the right answer?
Questions

Mathematics, 20.12.2019 00:31




English, 20.12.2019 00:31


Physics, 20.12.2019 00:31

History, 20.12.2019 00:31


Health, 20.12.2019 00:31

Chemistry, 20.12.2019 00:31


Mathematics, 20.12.2019 00:31

English, 20.12.2019 00:31

Business, 20.12.2019 00:31

English, 20.12.2019 00:31


Chemistry, 20.12.2019 00:31

Mathematics, 20.12.2019 00:31

Mathematics, 20.12.2019 00:31