A 50g block of aluminum at -100°C is submerged in 30g of water at 20°C.
Cwater = 4.184 J/gºC
...

Chemistry, 26.10.2021 19:30 lazavionadams81
A 50g block of aluminum at -100°C is submerged in 30g of water at 20°C.
Cwater = 4.184 J/gºC
AH tus water = 334 J/g
Cu = 0.89 J/gºC
What is the final temperature of both substances?
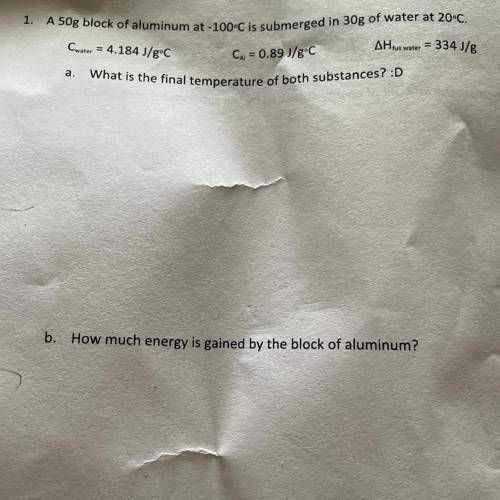

Answers: 1
Another question on Chemistry


Chemistry, 21.06.2019 19:20
What is the strongest intermolecular force between an nacl unit and an h2o molecule together in a solution? covalent bonding dipole-dipole force hydrogen bonding ion-dipole force
Answers: 1


Chemistry, 22.06.2019 22:30
Which process describes vaporization that takes place below the surface of a liquid? condensation melting boiling evaporation
Answers: 1
You know the right answer?
Questions

Mathematics, 29.05.2020 23:09

Chemistry, 29.05.2020 23:09

Geography, 29.05.2020 23:09




Mathematics, 29.05.2020 23:09

Biology, 29.05.2020 23:09









Mathematics, 29.05.2020 23:09






