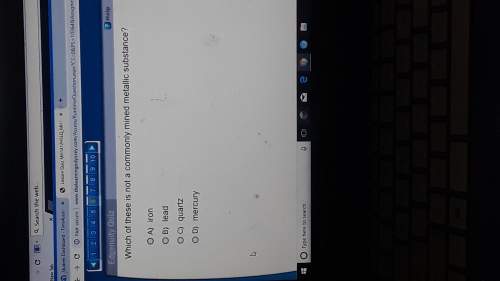
Chemistry, 26.11.2021 18:20 carsondelane13
Silver tarnishes as silver metal reacts with hydrogen sulfide, H2S, in the air. In this reaction, dark silver sulfide, Au2S, covers the surface of silver. When silver is polished, this coating of silver sulfide can be removed from the surface. This makes the silver shiny again. Enter the coefficients that balance the tarnishing reaction equation. (Type 1 for no coefficient. ) 2 silver dishes. Photo by Mgeurts Ag(s) H2S(g) → Ag2S(s) H2(g).

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 14:00
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1


Chemistry, 22.06.2019 19:50
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3
You know the right answer?
Silver tarnishes as silver metal reacts with hydrogen sulfide, H2S, in the air. In this reaction, da...
Questions


Mathematics, 24.09.2019 13:00

History, 24.09.2019 13:00


Mathematics, 24.09.2019 13:00

Mathematics, 24.09.2019 13:00



Mathematics, 24.09.2019 13:00

Mathematics, 24.09.2019 13:00

Spanish, 24.09.2019 13:00


English, 24.09.2019 13:00

Computers and Technology, 24.09.2019 13:00

Computers and Technology, 24.09.2019 13:00


Health, 24.09.2019 13:00

Mathematics, 24.09.2019 13:00


History, 24.09.2019 13:00