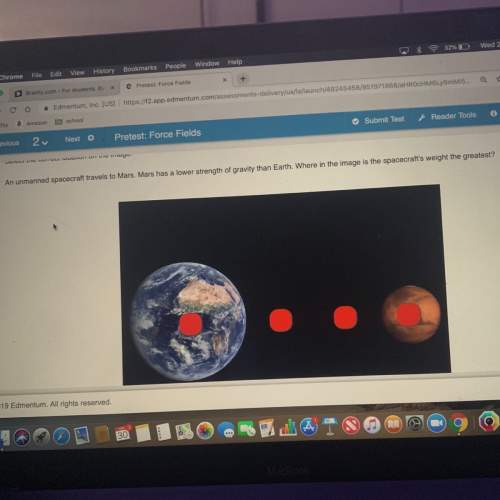
Chemistry, 29.11.2021 04:30 puremousetail
What is the pressure of 1.27 L of a gas at 288°C, if the gas had a volume of 875 ml at 145 kPa and 176°C?

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1
You know the right answer?
What is the pressure of 1.27 L of a gas at 288°C, if the gas had a volume of 875 ml at
145 kPa and...
Questions





Computers and Technology, 14.10.2019 21:30

Biology, 14.10.2019 21:30





Physics, 14.10.2019 21:30





English, 14.10.2019 21:30

Mathematics, 14.10.2019 21:30

History, 14.10.2019 21:30

Mathematics, 14.10.2019 21:30

Chemistry, 14.10.2019 21:30