
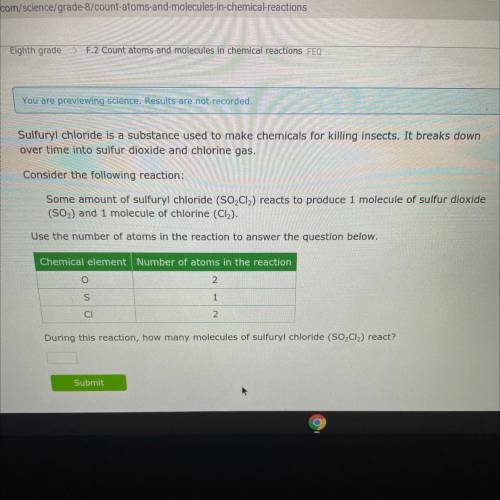
Chemistry, 30.11.2021 21:50 morgan3346
Sulfuryl chloride is a substance used to make chemicals for killing insects. It breaks down
over time into sulfur dioxide and chlorine gas.
Consider the following reaction:
Some amount of sulfuryl chloride (SO2Cl2) reacts to produce 1 molecule of sulfur dioxide
(S02) and 1 molecule of chlorine (CI).
Use the number of atoms in the reaction to answer the question below.
Chemical element Number of atoms in the reaction
0
2
s
1
CI
2
During this reaction, how many molecules of sulfuryl chloride (SO2Cl2) react?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:30
150.0 grams of asf3 were reacted with 180.0 g of ccl4 to produce ascl3 and ccl2f2. if the actual yield of ccl2f2 was 127 g, what is the percent yield?
Answers: 2

Chemistry, 22.06.2019 01:50
7. what temperature is need to just dissolve 50 g of nh4cl in 75 g of water? '
Answers: 1

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 07:30
Given that 1 mi = 1760 yd, determine what conver- sion factor is appropriate to convert 1849 yd to miles; to convert 2.781 mi to yards.
Answers: 2
You know the right answer?
Sulfuryl chloride is a substance used to make chemicals for killing insects. It breaks down
over t...
Questions

Physics, 12.08.2020 17:01



Biology, 12.08.2020 17:01

Mathematics, 12.08.2020 17:01




Mathematics, 12.08.2020 17:01

English, 12.08.2020 17:01

Health, 12.08.2020 17:01

Mathematics, 12.08.2020 17:01

Mathematics, 12.08.2020 17:01

History, 12.08.2020 17:01

Health, 12.08.2020 17:01

Mathematics, 12.08.2020 17:01


Spanish, 12.08.2020 17:01


Mathematics, 12.08.2020 17:01



