
Chemistry, 01.12.2021 08:30 vickydaiberg
Calculate the reaction rate if [Cl2] = 0.012M, [O2] = 0.250M and the rate constant is 2.54x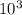
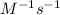 at 25C.
at 25C.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 23:00
What is the most common reason for matter changing its state?
Answers: 1

Chemistry, 23.06.2019 00:30
When did stem cell research become known ? who discovered stem cell? what experiments or studies have been conducted so far?
Answers: 3

Chemistry, 23.06.2019 04:00
Why must humans find substitutes for many minerals found on earth? (a) form at an extremely slow rate (b) controlled by other countries (c) too deep in the earth to collect
Answers: 1

Chemistry, 23.06.2019 07:30
Achemist at a pharmaceutical company is measuring equilibrium constants for reactions in which drug candidate molecules bind to a protein involved in cancer. the drug molecules bind the protein in a 1: 1 ratio to form a drug-protein complex. the protein concentration in aqueous solution at 25 ˚c is 1.74 x10-6 m . drug a is introduced into the protein solution at an initial concentration of 2.00 x10-6m. drug b is introduced into a separate, identical protein solution at an initial concentration of 2.00 x10-6m. at equilibrium, the drug a-protein solution has an a-protein complex concentration of 1.00 x10-6m, and the drug b solution has a b-protein complex concentration of 1.40 x10-6m.a. calculate the kc value for the a-protein binding reaction.b. calculate the kc value for the b-protein binding reaction.c. assuming that the drug that binds more strongly will be more effective, which drug is the better choice for further research?
Answers: 1
You know the right answer?
Calculate the reaction rate if [Cl2] = 0.012M, [O2] = 0.250M and the rate constant is 2.54x at 25C....
Questions

Mathematics, 26.03.2021 21:40

World Languages, 26.03.2021 21:40


History, 26.03.2021 21:40

Chemistry, 26.03.2021 21:40


Mathematics, 26.03.2021 21:40


Mathematics, 26.03.2021 21:40

Mathematics, 26.03.2021 21:40

History, 26.03.2021 21:40



Engineering, 26.03.2021 21:40

Physics, 26.03.2021 21:40







