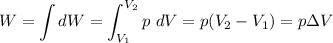Chemistry, 01.12.2021 23:50 lannor6586
How to calculate work done with change in volume and pressure.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2

Chemistry, 23.06.2019 07:40
Which of the following has expanded our knowledge of the universe beyond our solar system the most? a. manned space travel b. the hubble space telescope c. the pioneer and voyager missions d. the international space station
Answers: 3

Chemistry, 23.06.2019 13:00
Johnny's bakery has 30,900 grams of sugar. a recipe calls for 32 pounds of sugar to be used. how much sugar will be left over? (1 lb=453.59 g).
Answers: 2
You know the right answer?
How to calculate work done with change in volume and pressure....
Questions

Business, 05.10.2019 05:00



Mathematics, 05.10.2019 05:00

History, 05.10.2019 05:00

Mathematics, 05.10.2019 05:00

Mathematics, 05.10.2019 05:00

Mathematics, 05.10.2019 05:00


English, 05.10.2019 05:00


Social Studies, 05.10.2019 05:00


Mathematics, 05.10.2019 05:00

Mathematics, 05.10.2019 05:00

Computers and Technology, 05.10.2019 05:00


Mathematics, 05.10.2019 05:00

History, 05.10.2019 05:00

Chemistry, 05.10.2019 05:00