
Chemistry, 04.12.2021 20:50 LaToyaShante1228
Which list shows the phases of matter in order from the greatest average kinetic energy to the least average kinetic energy per particle?
A substance has a pH of 8.0. How would it be classified?
Accordingly to the gas laws, which variables are inversely proportional to each other (pressure, volume, temperature)?
part 2
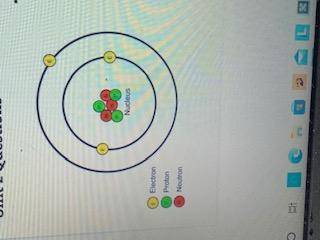
What is the name of this atom?
What is the atomic number?
What is the atomic mass?
What is the charge?


Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 23.06.2019 00:00
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1

Chemistry, 23.06.2019 00:20
What type of context clue you understand the meaning of quandary?
Answers: 3
You know the right answer?
Which list shows the phases of matter in order from the greatest average kinetic energy to the least...
Questions



Mathematics, 30.11.2020 19:40


Chemistry, 30.11.2020 19:40




Mathematics, 30.11.2020 19:40

Mathematics, 30.11.2020 19:40

Mathematics, 30.11.2020 19:40



History, 30.11.2020 19:40




Mathematics, 30.11.2020 19:40

Mathematics, 30.11.2020 19:40



