
Chemistry, 05.12.2021 22:10 jadeossowski3590
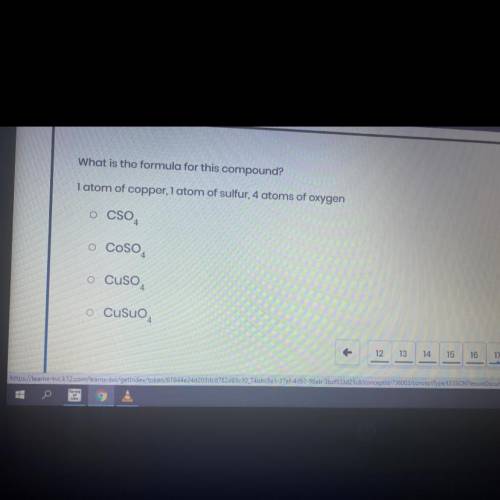
What is the formula for this compound? 1 atom of copper, 1 atom of sulfur, 4 atoms of oxygen

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 13:30
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3


Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1
You know the right answer?
What is the formula for this compound?
1 atom of copper, 1 atom of sulfur, 4 atoms of oxygen
Questions


English, 17.04.2021 22:40

Mathematics, 17.04.2021 22:40

Mathematics, 17.04.2021 22:40

Mathematics, 17.04.2021 22:40

Mathematics, 17.04.2021 22:40

Mathematics, 17.04.2021 22:40

Mathematics, 17.04.2021 22:40






English, 17.04.2021 22:40

Mathematics, 17.04.2021 22:40

Business, 17.04.2021 22:40

Mathematics, 17.04.2021 22:40





