
Chemistry, 06.12.2021 22:10 kyliegriffis
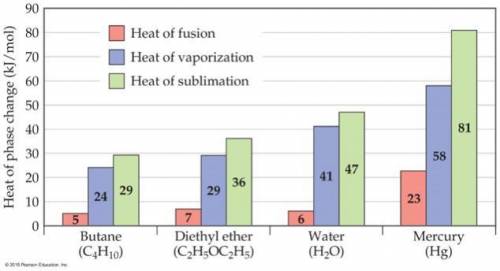
Consider the figure below. In the case of butane, diethyl ether, and water, the heat of vaporization is considerably larger than heat of fusion. Explain this phenomenon. Note that the nature of intermolecular interactions does not change through the phases because the molecules does not undergo chemical changes.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3


Chemistry, 22.06.2019 22:00
Plz ill give u brainliest which of the following steps is not likely to take place during cellular respiration? a.oxygen combines with carbon of simple sugar. b. energy molecule transfers energy to cells. d. energy is used up.
Answers: 3
You know the right answer?
Consider the figure below. In the case of butane, diethyl ether, and water, the heat of vaporization...
Questions

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01

Mathematics, 16.09.2020 04:01



