Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2
You know the right answer?
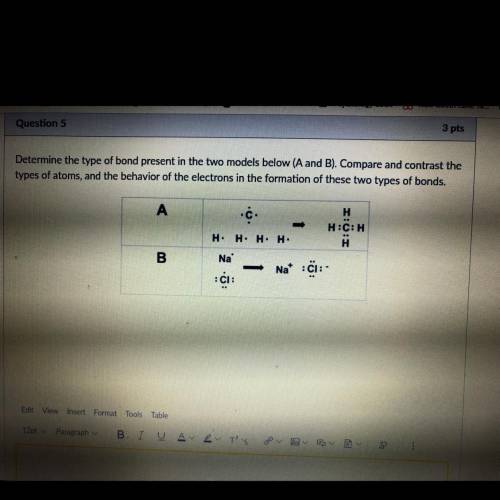
Determine the type of bond present in the two models below (A and B). Compare and contrast the
typ...
Questions









Computers and Technology, 01.12.2021 02:00

Mathematics, 01.12.2021 02:00

World Languages, 01.12.2021 02:00




SAT, 01.12.2021 02:00

Computers and Technology, 01.12.2021 02:00

Social Studies, 01.12.2021 02:00

Biology, 01.12.2021 02:00

Mathematics, 01.12.2021 02:00