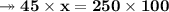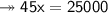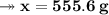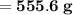Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 23.06.2019 04:00
How many liters of water can be produced from 5.0liters of butane gas at stp, assuming excess oxygen? c4h10(g) + 02(g) → co2 (e) + h2o (g)
Answers: 2

You know the right answer?
Knowing that the solubility of a salt at 80°C is 45g/100 g of H, O, calculate the mass of water requ...
Questions

Spanish, 06.12.2021 23:10


Mathematics, 06.12.2021 23:10

English, 06.12.2021 23:10



English, 06.12.2021 23:10


Computers and Technology, 06.12.2021 23:10


Mathematics, 06.12.2021 23:10

Mathematics, 06.12.2021 23:10

Arts, 06.12.2021 23:10


English, 06.12.2021 23:10

Social Studies, 06.12.2021 23:10

English, 06.12.2021 23:10



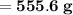 .
. 45g of salt
45g of salt  100g of H₂O
100g of H₂O