9
Ammonia is manufactured from nitrogen and hydrogen by the Haber process.
N2(g) + 3H2(g)2NH...

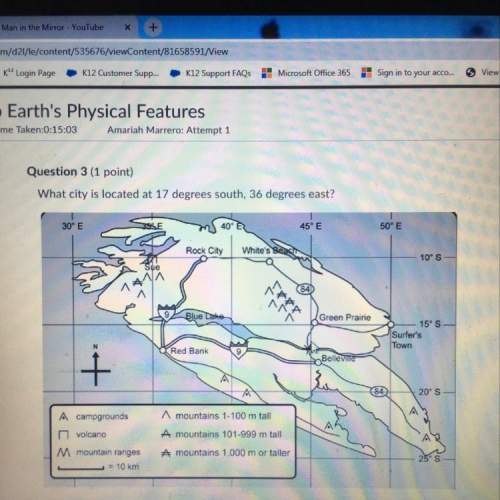
Chemistry, 26.12.2021 02:10 sweetbee633
9
Ammonia is manufactured from nitrogen and hydrogen by the Haber process.
N2(g) + 3H2(g)2NH3(g)
What is the percentage yield when 60 kg of ammonia is produced from 60 kg of hydrogen?
A
5.9%
B
17.6%
с
35.3%
D
50.0%

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
You know the right answer?
Questions






Mathematics, 02.04.2020 03:54



Mathematics, 02.04.2020 03:55

History, 02.04.2020 03:55


Computers and Technology, 02.04.2020 03:55

Mathematics, 02.04.2020 03:55




English, 02.04.2020 03:55


Mathematics, 02.04.2020 03:55

Mathematics, 02.04.2020 03:55