
PLEASE I NEED HELP ON THIS
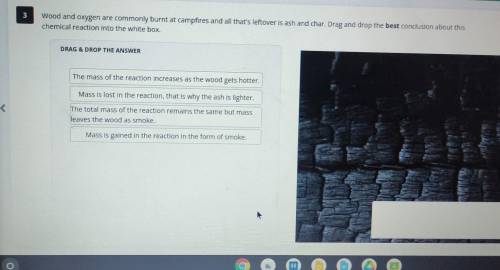
Wood and oxygen are commonly burnt at campfires and all that's leftover is ash and char. Drag and drop the best conclusion about this chemical reaction into the white box.
DRAG & DROP THE ANSWER
The mass of the reaction increases as the wood gets hotter.
Mass Is lost in the reaction, that is why the ash is lighter.
The total mass of the reaction remains the same but mass leaves the wood as smoke.
Mass is gained in the reaction in the form of smoke.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 23.06.2019 00:30
How can you write e method for the experiment of separating sand from water by filtration process? 1-materials 2-steps 3-conclusion also the same for the separating process of water and salt by filtration or distillation. quick because i need to finish my hw
Answers: 2

Chemistry, 23.06.2019 02:00
Pinene is an unsaturated hydrocarbon found in pine resin. if pinene has m+ = 136 and contains 1 double bond(s) and 2 ring(s); what is its molecular formula? enter the formula in the form ch first, then all other atoms in alphabetical order; do not use subscripts. the formula is case-sensitive
Answers: 3

Chemistry, 23.06.2019 03:00
The size (radius) of an oxygen molecule is about 2.0 ×10−10m. make a rough estimate of the pressure at which the finite volume of the molecules should cause noticeable deviations from ideal-gas behavior at ordinary temperatures (t= 300k ). assume that deviatons would be noticeable when volume of the gas per molecule equals the volume of the molecule itself.
Answers: 3
You know the right answer?
PLEASE I NEED HELP ON THIS
Wood and oxygen are commonly burnt at campfires and all that's leftover...
Questions

English, 20.10.2019 14:30



History, 20.10.2019 14:30

Mathematics, 20.10.2019 14:30

Mathematics, 20.10.2019 14:30

Mathematics, 20.10.2019 14:30

Mathematics, 20.10.2019 14:30

Mathematics, 20.10.2019 14:30


Mathematics, 20.10.2019 14:30


History, 20.10.2019 14:30



History, 20.10.2019 14:30


History, 20.10.2019 14:30

History, 20.10.2019 14:30



