Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2


Chemistry, 23.06.2019 05:00
How is electrolysis most commonly used to produce an energy source? a - splitting water molecules produces oxygen, which organisms breathe to fuel their bodies. b - splitting water molecules produces hydrogen gas, which is used to power machines through hydrogen fuel cells. c - splitting carbon dioxide molecules produces coal, a form of carbon that can be burned to produce heat. d - splitting carbon dioxide molecules produces natural gas, which can be burned to generate electricity in power plants.
Answers: 1
You know the right answer?
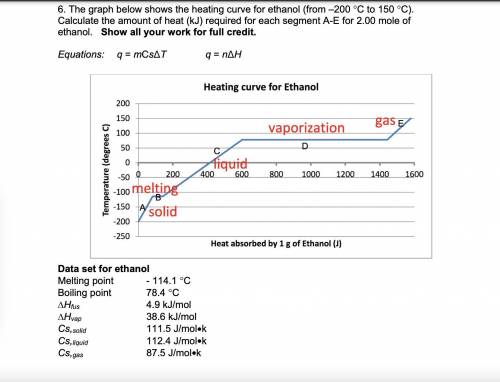
6. The graph below shows the heating curve for ethanol (from –200C to 150C). Calculate the amount of...
Questions

English, 21.09.2019 08:30

History, 21.09.2019 08:30

Chemistry, 21.09.2019 08:30

Mathematics, 21.09.2019 08:30



Geography, 21.09.2019 08:30

Mathematics, 21.09.2019 08:30

Mathematics, 21.09.2019 08:30

History, 21.09.2019 08:30

Mathematics, 21.09.2019 08:30

English, 21.09.2019 08:30


Social Studies, 21.09.2019 08:30





Health, 21.09.2019 08:30

Mathematics, 21.09.2019 08:30