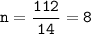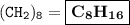Chemistry, 08.02.2022 14:00 tatilynnsoto17
A compound with the empirical formula CH2 was found to have a molar mass of approximately 112 g. Write the molecular formula of the compound.
2Points
Show all your work. Please use correct formatting for subscripts and exponents. The math formula editor makes it easier to show work.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1

Chemistry, 22.06.2019 18:30
Which of the following nuclei would be the least stable a 2 protons, 2 neutrons b 1 proton 1 neutron c 1 proton 3 neutrons d 1 proton 2 neutrons
Answers: 3

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

Chemistry, 22.06.2019 22:30
What must be in balance for temperatures to remain constant?
Answers: 1
You know the right answer?
A compound with the empirical formula CH2 was found to have a molar mass of approximately 112 g. Wri...
Questions

Mathematics, 16.10.2019 02:00




Physics, 16.10.2019 02:00

History, 16.10.2019 02:00

History, 16.10.2019 02:00



Mathematics, 16.10.2019 02:00


History, 16.10.2019 02:00


Mathematics, 16.10.2019 02:00



Mathematics, 16.10.2019 02:00

Mathematics, 16.10.2019 02:00


History, 16.10.2019 02:00