
Chemistry, 13.02.2022 04:40 DesireeGuzman5213
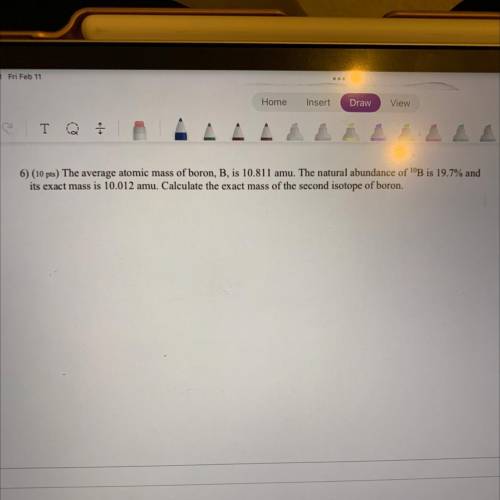
6) The average atomic mass of boron, B, is 10.811 amu. The natural abundance of 10B is 19.7% and it’s exact mass is 10.012 amu. Calculate the exact mass of the second isotope of boron.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
The agent of mechanical weathering in which rock is worn away by the grinding action of other rock particles is call
Answers: 1

Chemistry, 22.06.2019 05:30
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2


Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
You know the right answer?
6) The average atomic mass of boron, B, is 10.811 amu. The natural abundance of 10B is 19.7% and it’...
Questions




English, 03.03.2020 03:34

Social Studies, 03.03.2020 03:34

















