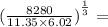A =
What is the answer?...

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 02:10
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3


Chemistry, 22.06.2019 04:30
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2
You know the right answer?
Questions

History, 03.03.2020 08:22

Physics, 03.03.2020 08:22

Mathematics, 03.03.2020 08:22


English, 03.03.2020 08:23

Biology, 03.03.2020 08:23

History, 03.03.2020 08:24

Social Studies, 03.03.2020 08:25


Mathematics, 03.03.2020 08:25

Chemistry, 03.03.2020 08:26

Mathematics, 03.03.2020 08:26

Health, 03.03.2020 08:27


Mathematics, 03.03.2020 08:35


Mathematics, 03.03.2020 08:35

English, 03.03.2020 08:35

Mathematics, 03.03.2020 08:36