
Chemistry, 16.03.2022 20:00 kierafisher05
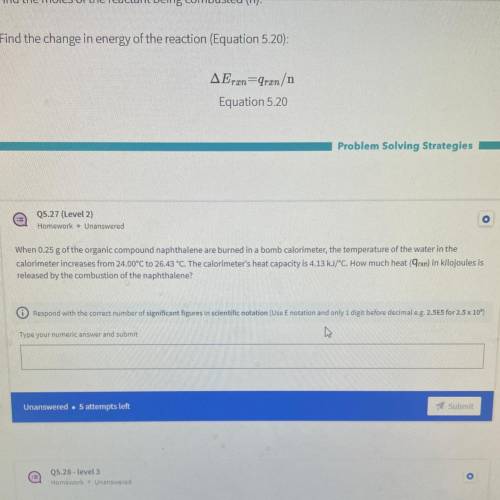
When 0.25 g of the organic compound naphthalene are burned in a bomb calorimeter, the temperature of the water in the
calorimeter increases from 24.00°C to 26.43 °C. The calorimeter's heat capacity is 4.13 kJ/°C. How much heat (9rxn) in kilojoules is
released by the combustion of the naphthalene?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:10
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1
You know the right answer?
When 0.25 g of the organic compound naphthalene are burned in a bomb calorimeter, the temperature of...
Questions


Mathematics, 31.01.2020 17:57

English, 31.01.2020 17:57



Mathematics, 31.01.2020 17:57


Mathematics, 31.01.2020 17:57








Social Studies, 31.01.2020 17:58

Social Studies, 31.01.2020 17:58


Mathematics, 31.01.2020 17:58

Mathematics, 31.01.2020 17:58



