
Chemistry, 30.06.2019 11:40 ErrorNameTaken505
Why is respiration an exothermic reaction? a large amount of energy is released into the surroundings. the heat content, q, of the reaction is positive. a large amount of energy is required to activate the reaction. there is no exchange of energy between the system and the surrounding.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 06:00
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3


Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
You know the right answer?
Why is respiration an exothermic reaction? a large amount of energy is released into the surroundin...
Questions



Chemistry, 26.02.2021 22:50


Mathematics, 26.02.2021 22:50


Biology, 26.02.2021 22:50



Mathematics, 26.02.2021 22:50


English, 26.02.2021 22:50

Mathematics, 26.02.2021 22:50



Mathematics, 26.02.2021 22:50


Mathematics, 26.02.2021 22:50


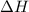 which is difference between energy of products and energy of reactants come out to be positive.
which is difference between energy of products and energy of reactants come out to be positive.


