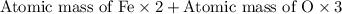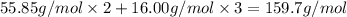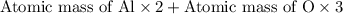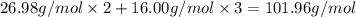Chemistry, 03.07.2019 06:00 HannaTheGurls
You can use mole ratios and molar mass to determine the masses of substances involved in a chemical reaction. using the periodic table, calculate the molar masses of iron oxide (fe2o3) and aluminum oxide (al2o3). give your answer to four significant figures.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Each of the following compounds contains a metal that can exhibit more than one ionic charge. provide systematic names for each of these compounds. (a) cr(clo3)6 (b) mo(cn)6 (c) cr2(so3)3 (d) v(clo2)2 (e) v(cn)5 (f) os(clo2)4
Answers: 3

Chemistry, 22.06.2019 01:00
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1
You know the right answer?
You can use mole ratios and molar mass to determine the masses of substances involved in a chemical...
Questions

Chemistry, 26.07.2019 21:30

Social Studies, 26.07.2019 21:30











History, 26.07.2019 21:30



Mathematics, 26.07.2019 21:30

Chemistry, 26.07.2019 21:30

Social Studies, 26.07.2019 21:30


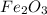 is 159.7 g/mol.
is 159.7 g/mol.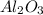 is 101.96 g/mol.
is 101.96 g/mol.