Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 08:30
How does the principle of electromagnetism explain the interaction between earth’s magnetic field and the solar wind?
Answers: 1


Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
You know the right answer?
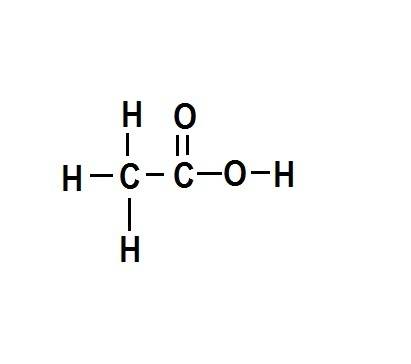
Why aren't any of the pictures a good representation of hc2h3o2(aq)?...
Questions

Mathematics, 07.02.2022 01:00



Mathematics, 07.02.2022 01:00


Spanish, 07.02.2022 01:00

Mathematics, 07.02.2022 01:00



Business, 07.02.2022 01:00

Mathematics, 07.02.2022 01:00



History, 07.02.2022 01:00



Mathematics, 07.02.2022 01:00



Business, 07.02.2022 01:00