
Chemistry, 09.07.2019 16:30 RandomLollipop
Nitroglycerin (c3h5n3o9) is a powerful explosive. its decomposition may be represented by 4c3h5n3o9 → 6n2 12co2 10h2o o2 this reaction generates a large amount of heat and gaseous products. it is the sudden formation of these gases, together with their rapid expansion, that produces the explosion. (a) what is the maximum amount of o2 in grams that can be obtained from 4.50 × 102 g of nitroglycerin

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1

Chemistry, 23.06.2019 02:00
The bohr model of the atom explained why emission spectra are discrete. it could also be used to explain the photoelectric effect. which is a correct explanation of the photoelectric effect according to the model?
Answers: 3

Chemistry, 23.06.2019 03:50
Which best describes the activation energy of a chemical reaction? a. the combined energy of all the reactants b. the amount of energy required for a reaction to occur c. the difference in energy between products and reactants d. the potential energy stored in the bonds of reactants and products
Answers: 1
You know the right answer?
Nitroglycerin (c3h5n3o9) is a powerful explosive. its decomposition may be represented by 4c3h5n3o9...
Questions


Engineering, 14.12.2020 09:30

Mathematics, 14.12.2020 09:30

Mathematics, 14.12.2020 09:30

Mathematics, 14.12.2020 09:30


History, 14.12.2020 09:30

Spanish, 14.12.2020 09:30

Chemistry, 14.12.2020 09:30


Mathematics, 14.12.2020 09:30



Chemistry, 14.12.2020 09:30


Mathematics, 14.12.2020 09:30




Mathematics, 14.12.2020 09:30

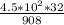 = 15.86 g of O₂
= 15.86 g of O₂

