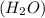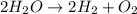Chemistry, 13.07.2019 23:30 lizbethreyes5725
Which is the most accurate description of compounds? a. they are formed by elements that join in any given mass ratio. b. they have the same properties as the elements that make them up. c. they have a unique set of properties that can be used as identifiers. d. they cannot be broken down into the elements that form them through chemical changes.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 4.20 mol fe and 6.70 mol nio(oh) react?
Answers: 3

Chemistry, 23.06.2019 11:50
Achemist needs to prepare a buffer solution of ph 8.80. what molarity of nh3 (pkb = 4.75) is required to produce the buffer solution if the (nh4)2so4 in the solution is 1.8 m?
Answers: 1
You know the right answer?
Which is the most accurate description of compounds? a. they are formed by elements that join in a...
Questions

Advanced Placement (AP), 23.08.2019 05:20


Computers and Technology, 23.08.2019 05:20


Mathematics, 23.08.2019 05:20



Mathematics, 23.08.2019 05:20






Mathematics, 23.08.2019 05:20


Mathematics, 23.08.2019 05:20


History, 23.08.2019 05:20