
Chemistry, 18.07.2019 10:10 keishah577
The hydrogen gas formed in a chemical reaction is collected over water at 30.0 ∘c at a total pressure of 734 mmhg . part a what is the partial pressure of the hydrogen gas collected in this way?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5 m hcl? show all of the work needed to solve this problem. mg (s) + 2hcl (aq) → mgcl2 (aq) + h2 (g)
Answers: 3

Chemistry, 23.06.2019 02:00
Which would freeze at a higher temperature: the great salt lake or lake tahoe? a. lake tahoe would freeze at a higher temperature. b. the great salt lake would freeze at a higher temperature. c. both lakes would freeze at the same temperature.
Answers: 2

Chemistry, 23.06.2019 08:30
According to the passage, which of these is true about gray water systems? a) gray water systems use plants that require less water. eliminate b) gray water systems require the use of less fossil fuels. c) gray water systems reduce the amount of fresh water used. d) gray water systems reduce the amount water used by shower heads.
Answers: 1

Chemistry, 23.06.2019 10:30
Ireally need ! calcium metal reacts with a potassium chloride solution to form calcium chloride and potassium ions. balance this reaction. (s) + (aq) → cacl2(s) + +(aq) a) 1, 2, 1, 2 b) 1, 2, 1, 1 c) 1, 1, 1, 1 d) 2, 1, 2, 1
Answers: 1
You know the right answer?
The hydrogen gas formed in a chemical reaction is collected over water at 30.0 ∘c at a total pressur...
Questions





Advanced Placement (AP), 21.06.2019 16:00



Mathematics, 21.06.2019 16:00






Mathematics, 21.06.2019 16:00


Chemistry, 21.06.2019 16:00


Chemistry, 21.06.2019 16:00

History, 21.06.2019 16:00

History, 21.06.2019 16:00

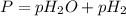
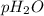 = partial pressure of water
= partial pressure of water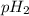 = partial pressure of hydrogen
= partial pressure of hydrogen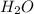 at 30°C is 31.8 mmHg
at 30°C is 31.8 mmHg

