
Chemistry, 21.07.2019 03:10 lizzyhearts
Subject science! distillation a. is the best method for separating a solid and a liquid b. uses boiling points to separate different liquids c. separates substances based on particle size d. causes a chemical change

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1

Chemistry, 22.06.2019 22:00
If a solution contains 3 moles/liter of sodium chloride (nacl, made of sodium ions and chloride ions), what is the osmolarity of this solution
Answers: 3

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
Subject science! distillation a. is the best method for separating a solid and a liquid b. uses bo...
Questions






Mathematics, 18.07.2019 20:30



Chemistry, 18.07.2019 20:30

Mathematics, 18.07.2019 20:30



Mathematics, 18.07.2019 20:30



Mathematics, 18.07.2019 20:30

Mathematics, 18.07.2019 20:30

Mathematics, 18.07.2019 20:30


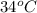 and dioxane (boiling point
and dioxane (boiling point 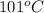 ) can be separated out by distillation.
) can be separated out by distillation.

