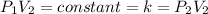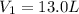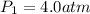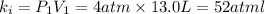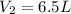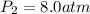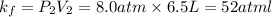Chemistry, 23.07.2019 09:40 catalanb474
Acertain gas is present in a 13.0 l cylinder at 4.0 atm pressure. if the pressure is increased to 8.0 atm , the volume of the gas decreases to 6.5 l . find the two constants ki, the initial value of k, and kf, the final value of k, to verify whether the gas obeys boyle's law. express your answers to two significant figures separated by a comma.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 02:30
Needthe meter is the standard unit for: 1) height 2) length 3) weight 4) mass
Answers: 3

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1
You know the right answer?
Acertain gas is present in a 13.0 l cylinder at 4.0 atm pressure. if the pressure is increased to 8....
Questions

English, 30.01.2020 19:45


Mathematics, 30.01.2020 19:45





Mathematics, 30.01.2020 19:45

Mathematics, 30.01.2020 19:45





Mathematics, 30.01.2020 19:45

History, 30.01.2020 19:45

History, 30.01.2020 19:45

Mathematics, 30.01.2020 19:45

Mathematics, 30.01.2020 19:45

Social Studies, 30.01.2020 19:45

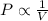 (At constant temperature and number of moles)
(At constant temperature and number of moles)