
Chemistry, 24.07.2019 10:30 graymonky12
Balance the following equation. then determine the ratio for the products kcl and o2 generated during the decomposition of potassium chlorate. kclo3 kcl + o2 1: 1 2: 2 4: 3 2: 3 3: 2

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 23.06.2019 00:30
Which radioisotope is used to date fossils? a. oxygen-16 b. carbon-14 c. uranium-238 d. carbon-12
Answers: 2

Chemistry, 23.06.2019 05:30
Awhite powder is added to a solution. the images show observations made before the powder is added, just after the powder has been added, and a little while later. (the liquid in the small beaker is phenol red solution.) what evidence shows that a chemical change has taken place?
Answers: 1

Chemistry, 23.06.2019 05:30
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table? atom p has an estimated zeff of 7 and is therefore to the left of atom q, which has a zeff of 6. atom p has an estimated zeff of 7 and is therefore to the right of atom q, which has a zeff of 6. atom p has an estimated zeff of 5 and is therefore below atom q, which has a zeff of 4. atom p has an estimated zeff of 5 and is therefore above atom q, which has a zeff of 4.
Answers: 3
You know the right answer?
Balance the following equation. then determine the ratio for the products kcl and o2 generated durin...
Questions

Mathematics, 03.07.2019 22:00



Chemistry, 03.07.2019 22:00



Mathematics, 03.07.2019 22:00



History, 03.07.2019 22:00

Mathematics, 03.07.2019 22:00




Social Studies, 03.07.2019 22:00

Mathematics, 03.07.2019 22:00

History, 03.07.2019 22:00

Mathematics, 03.07.2019 22:00

Mathematics, 03.07.2019 22:00

Mathematics, 03.07.2019 22:00

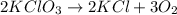
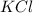 and 3 moles of
and 3 moles of 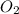 .
.

