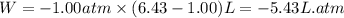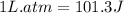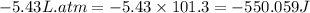One mole of an ideal gas is expanded from a volume of 1.00 liter to a volume of 6.43 liters against a constant external pressure of 1.00 atm. how much work (in joules) is performed on the surroundings? ignore significant figures for this problem. (t = 300 k; 1 l·atm = 101.3 j)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 15:00
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
One mole of an ideal gas is expanded from a volume of 1.00 liter to a volume of 6.43 liters against...
Questions

English, 20.08.2019 15:50


Chemistry, 20.08.2019 15:50


History, 20.08.2019 15:50

History, 20.08.2019 15:50

History, 20.08.2019 15:50

English, 20.08.2019 15:50





Biology, 20.08.2019 15:50

Biology, 20.08.2019 15:50


Computers and Technology, 20.08.2019 15:50

English, 20.08.2019 15:50

Mathematics, 20.08.2019 15:50

Social Studies, 20.08.2019 15:50

History, 20.08.2019 15:50

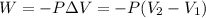
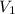 = initial volume = 1.00 L
= initial volume = 1.00 L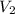 = final volume = 6.43 L
= final volume = 6.43 L