

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1

Chemistry, 23.06.2019 00:10
Covalent compounds: mastery test select the correct answer what is formed when atoms join together with a covalent bond? a. an ion b. a molecule c. a neutral atom d. a noble gas
Answers: 3
You know the right answer?
Which of the following indicates that an exothermic reaction has occurred? temperature increases he...
Questions

Mathematics, 27.01.2021 16:00


History, 27.01.2021 16:00


English, 27.01.2021 16:00

Chemistry, 27.01.2021 16:00

Mathematics, 27.01.2021 16:00

English, 27.01.2021 16:00

Advanced Placement (AP), 27.01.2021 16:00

Advanced Placement (AP), 27.01.2021 16:00



Chemistry, 27.01.2021 16:00

English, 27.01.2021 16:00

Mathematics, 27.01.2021 16:00


Physics, 27.01.2021 16:00


Mathematics, 27.01.2021 16:00

Mathematics, 27.01.2021 16:00

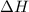 is positive for an endothermic process. This also means that a decrease in temperature occurs with absorption of heat.
is positive for an endothermic process. This also means that a decrease in temperature occurs with absorption of heat.

