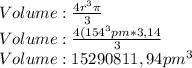Chemistry, 01.08.2019 05:00 Tyrant4life
Given the atomic radius of neon, 0.69 å, and knowing that a sphere has a volume of 4πr3/3, calculate the fraction of space that ne atoms occupy in a sample of neon at stp.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3


Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1

Chemistry, 22.06.2019 04:00
Two nitro no2 groups are chemically bonded to a patch of surface. they can't move to another location on the surface, but they can rotate (see sketch at right). it turns out that the amount of rotational kinetic energy each no2 group can have is required to be a multiple of ε, where =ε×1.010−24 j. in other words, each no2 group could have ε of rotational kinetic energy, or 2ε, or 3ε, and so forth — but it cannot have just any old amount of rotational kinetic energy. suppose the total rotational kinetic energy in this system is initially known to be 32ε. then, some heat is removed from the system, and the total rotational kinetic energy falls to 18ε. calculate the change in entropy. round your answer to 3 significant digits, and be sure it has the correct unit symbol.
Answers: 2
You know the right answer?
Given the atomic radius of neon, 0.69 å, and knowing that a sphere has a volume of 4πr3/3, calculate...
Questions

Mathematics, 24.11.2020 04:00

Mathematics, 24.11.2020 04:00

History, 24.11.2020 04:00

Mathematics, 24.11.2020 04:00



Social Studies, 24.11.2020 04:00

Mathematics, 24.11.2020 04:00




Mathematics, 24.11.2020 04:00




Mathematics, 24.11.2020 04:00


Computers and Technology, 24.11.2020 04:00


Mathematics, 24.11.2020 04:00