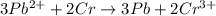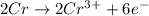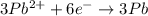Given the balanced ionic equation:
3pb2+ + 2cr ==> 3pb + 2cr3+
what is the number o...

Chemistry, 20.08.2019 11:30 mathman783
Given the balanced ionic equation:
3pb2+ + 2cr ==> 3pb + 2cr3+
what is the number of moles of electrons gained by 3.0 moles of lead ions?
(1) 5.0 mol (3)3.0 mol
(2) 2.0 mol (4) 6.0mol

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:00
An example of technology is the a. addition of a side group to an organic molecule during synthesis. b. use of a new antibiotic to fight an infection. c. measurement of iron concentration in a water sample. d. study of atomic fusion reactions
Answers: 3

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 23.06.2019 02:00
What is the source of continuous heat and energy that we receive from the sun
Answers: 2

Chemistry, 23.06.2019 10:30
How is it possible for someone to put an ear to a wall and hear someone in the next room? a.sound waves can travel though solids. b.the waves travel from room to room via air. c.there must be some air in the wall so the sound can travel through it. d.sound waves change to electromagnetic waves and then back again.
Answers: 1
You know the right answer?
Questions

History, 05.05.2020 17:57

Social Studies, 05.05.2020 17:57



Business, 05.05.2020 17:57

Mathematics, 05.05.2020 17:57

Mathematics, 05.05.2020 17:57


Mathematics, 05.05.2020 17:57

Biology, 05.05.2020 17:57



Mathematics, 05.05.2020 17:57