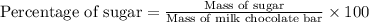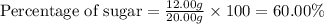Chemistry, 04.08.2019 03:00 bambamshortymacc
Given that a 20.00 g milk chocolate bar contains 12.00 g of sugar, calculate the percentage of sugar present in 20.00 g of milk chocolate bar keeping in mind that the answer should have four significant figures

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:20
Identify and describe the three ways that mutations affect organisms.
Answers: 1

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 18:30
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1
You know the right answer?
Given that a 20.00 g milk chocolate bar contains 12.00 g of sugar, calculate the percentage of sugar...
Questions


English, 19.07.2019 08:00


Mathematics, 19.07.2019 08:00




History, 19.07.2019 08:00

Geography, 19.07.2019 08:00


Mathematics, 19.07.2019 08:00

SAT, 19.07.2019 08:00


History, 19.07.2019 08:00

Mathematics, 19.07.2019 08:00


Social Studies, 19.07.2019 08:00


English, 19.07.2019 08:00