
Chemistry, 04.08.2019 00:30 bullockarwen
Magnesium burns in air with a dazzling brilliance to produce magnesium oxide: 2mg(s) + o2(g) → 2mgo(s) how many moles of o2 are consumed when 0.550 mol of magnesium burns?

Answers: 1
Another question on Chemistry

Chemistry, 23.06.2019 00:00
What is the pressure of 0.500 moles of carbon dioxide gas in a 2.5 l tank and at a temperature of 301 k? (r=0.0821 l·atm/mol·k) 3.08 atm 1.2 atm 0.23 atm 4.01 atm 4.94 atm
Answers: 1

Chemistry, 23.06.2019 07:50
Asolution is produced in which water is the solvent and there are four solutes. which of the solutes can dissolve better if the solution is heated?
Answers: 1

Chemistry, 23.06.2019 11:20
Which of the following is a pure substance? airbloodcopperwood
Answers: 2

Chemistry, 23.06.2019 12:00
Jill is pushing a box across the floor. which represents the upward force perpendicular to the floor? a) fp b) ff c) fn d) fg
Answers: 1
You know the right answer?
Magnesium burns in air with a dazzling brilliance to produce magnesium oxide: 2mg(s) + o2(g) → 2mgo...
Questions

Mathematics, 13.05.2021 16:20

Biology, 13.05.2021 16:20



Mathematics, 13.05.2021 16:20


Mathematics, 13.05.2021 16:20

Physics, 13.05.2021 16:20

Spanish, 13.05.2021 16:20

Mathematics, 13.05.2021 16:20

Chemistry, 13.05.2021 16:20

Mathematics, 13.05.2021 16:20



Mathematics, 13.05.2021 16:20


Mathematics, 13.05.2021 16:20



Mathematics, 13.05.2021 16:20

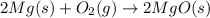
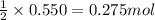 of oxygen gas.
of oxygen gas.

