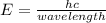Chemistry, 03.08.2019 17:30 rachelreed
When an electron of an atom falls from a higher energy level to the ground state, the atom loses 9.4145 x 10-25 joules of energy. what is the wavelength of the radiation emitted as a result of this transition? (planck’s constant is 6.626 x 10-34 joule seconds; the speed of light is 2.998 x 108m/s)?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2

Chemistry, 22.06.2019 13:00
Asubstance is a good conductor of electricity which of the following best explains a probable position of the substance in a periodic table
Answers: 3

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2
You know the right answer?
When an electron of an atom falls from a higher energy level to the ground state, the atom loses 9.4...
Questions



Biology, 18.07.2019 10:00

Mathematics, 18.07.2019 10:00


History, 18.07.2019 10:00




Mathematics, 18.07.2019 10:00


History, 18.07.2019 10:00


Mathematics, 18.07.2019 10:00



History, 18.07.2019 10:00


Mathematics, 18.07.2019 10:00