
Me i beg you
the average kinetic energy of 1 mole of a gas at -32 degrees celsius is:
3.01 x 103 j
3.99 x 102 j
-3.99 x 102 j
3.80 x 103 j
the relationship between volume and temperature of a gas, when pressure and moles of a gas are held constant, is: v*t = k.
true
false
the relationship between moles and volume, when pressure and temperature of a gas are held constant, is: v/n = k. we could say then, that:
if the moles of gas are tripled, the volume must also triple.
if the moles of gas are halved, the volume must be doubled.
if the moles of gas are doubled, the volume must be halved.
if the moles of gas are doubled, the volume is unaffected.
if the temperature and volume of a gas are held constant, an increase in pressure would most likely be caused by an increase in the number of moles of gas.
true
false
if the vapor pressure of a liquid is less than the atmospheric pressure, the liquid will not boil.
true
false
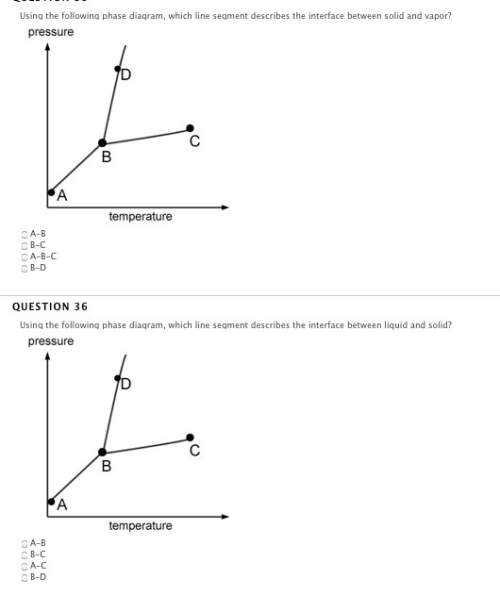

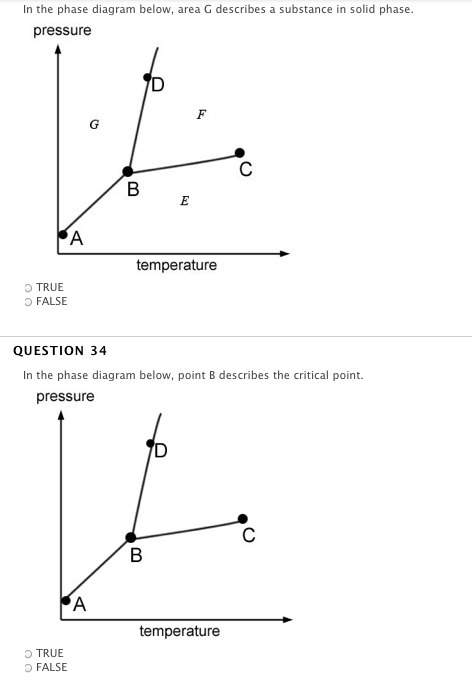
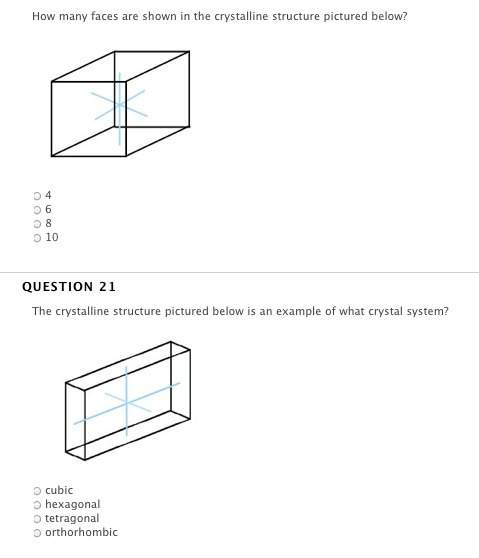

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 08:40
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2

Chemistry, 22.06.2019 11:40
Consider this equilibrium: n29) + o2(g) + 2no(c).nitrogen gas and oxygen gas react when placed in a closed container. as the reaction proceeds towards equilibrium, what happens to the rate of thereverse reaction?
Answers: 1

Chemistry, 22.06.2019 13:30
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2
You know the right answer?
Me i beg you
the average kinetic energy of 1 mole of a gas at -32 degrees celsius is: <...
the average kinetic energy of 1 mole of a gas at -32 degrees celsius is: <...
Questions


Mathematics, 30.08.2019 19:00

Chemistry, 30.08.2019 19:00





Mathematics, 30.08.2019 19:00

Mathematics, 30.08.2019 19:00

Mathematics, 30.08.2019 19:00




Social Studies, 30.08.2019 19:00




Biology, 30.08.2019 19:00

Chemistry, 30.08.2019 19:00

Biology, 30.08.2019 19:00



