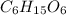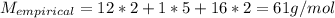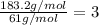Chemistry, 30.07.2019 02:00 sunshinekisses
Determine the molecular formula of a compound that has a molar mass of 183.2 g/mol and an empirical formula of c2h5o2.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3

Chemistry, 22.06.2019 21:50
If e is the symbol for an element, which two of the following symbols represent isotopes of the same element? 1. e2. e3. ea.1 and 2c.1 and 4b.3 and 4d.2 and 3
Answers: 2

Chemistry, 22.06.2019 22:30
Consider a culture medium on which only gram-positive organisms such as staphylococcus aureus colonies can grow due to an elevated nacl level. a yellow halo surrounds the growth, indicating the bacterium fermented a sugar in the medium, decreasing the ph as a result and changing the color of a ph indicator chemical. this type of medium would be referred to as a differential and enrichment culture.
Answers: 2

Chemistry, 23.06.2019 05:10
Will mark as brainliest ! how many grams of iron metal do you expect to be produced when 245 grams of an 80.5 percent by mass iron (ii) nitrate solution react with excess aluminum metal? show all work needed to solve this problem!
Answers: 1
You know the right answer?
Determine the molecular formula of a compound that has a molar mass of 183.2 g/mol and an empirical...
Questions


Mathematics, 16.12.2020 19:40


Arts, 16.12.2020 19:40


Mathematics, 16.12.2020 19:40

English, 16.12.2020 19:40




Mathematics, 16.12.2020 19:40



English, 16.12.2020 19:40


Mathematics, 16.12.2020 19:40

Mathematics, 16.12.2020 19:40


Mathematics, 16.12.2020 19:40

Mathematics, 16.12.2020 19:40