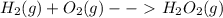Chemistry, 30.07.2019 02:00 ddmoorehouseov75lc
Write a balanced chemical equation depicting the formation of one mole of h2o2(g) from its elements in their standard states. express your answer as a chemical equation. identify all of the phases in your answer.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Which is true of the reactants in this displacement reaction? fe + 2hcl fecl2 + h2 a. the reactants are located to the left of the arrow in the chemical equation. b. the reactants contain 1 iron atom, 2 hydrogen atoms, and 1 chlorine atom. c. the reactants are the atoms, molecules, or compounds formed in the reaction. d. the reactants have the same physical and chemical properties as the products.
Answers: 1

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3


Chemistry, 22.06.2019 10:10
When electrolyzing copper (ll) chloride, what reaction takes place at the anode? what reaction takes place at the cathode?
Answers: 1
You know the right answer?
Write a balanced chemical equation depicting the formation of one mole of h2o2(g) from its elements...
Questions







Biology, 26.05.2021 01:00

Mathematics, 26.05.2021 01:00


History, 26.05.2021 01:00


Mathematics, 26.05.2021 01:00






Chemistry, 26.05.2021 01:00

Mathematics, 26.05.2021 01:00

Social Studies, 26.05.2021 01:00