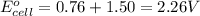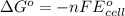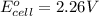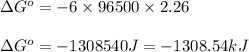Chemistry, 30.07.2019 01:30 wesleygrimes0
Calculate the standard free-energy change for the following reaction at 25 c. standard reduction potentials can be found here. 2au^3+ (aq) + 3zn (s) < --> 2au (s) + 3zn^2+(aq) can someone me with this?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1

Chemistry, 22.06.2019 22:30
Astudent pours 10.0 g of salt into a container of water and observes the amount of time it takes for the salt to dissolve. she then repeats the process using the same amounts of salt and water but this time she slowly stirs the mixture while it is dissolving. the student performs the experiment one more time but this time she stirs the mixture rapidly. the dependent variable in this experiment is: time for salt to dissolve speed of stirring amount of water mass of salt
Answers: 1

Chemistry, 23.06.2019 00:30
Quickly what are the following of organisms that existed over a wide area but only for a limited time period called a.soft fossils b.mold fossils c.index fossils d.trace fossils
Answers: 1
You know the right answer?
Calculate the standard free-energy change for the following reaction at 25 c. standard reduction pot...
Questions




History, 12.10.2019 07:20






History, 12.10.2019 07:20


History, 12.10.2019 07:20

Biology, 12.10.2019 07:20


Health, 12.10.2019 07:20

Biology, 12.10.2019 07:20

Computers and Technology, 12.10.2019 07:20



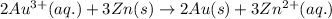
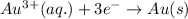 × 2
× 2 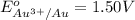
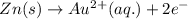 × 3
× 3 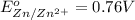
 , we use the equation:
, we use the equation: