
Chemistry, 29.07.2019 13:30 aurelio1121
Radon (rn) is the heaviest and the only radioactive member of group 8a(18), the noble gases. it is a product of the disintegration of heavier radioactive nuclei found in minute concentrations in many common rocks used for building and construction. in recent years, health concerns about the cancers caused from inhaled residential radon have grown. if 1.00 × 1015 atoms of radium (ra) produce an average of 1.373 × 104 atoms of rn per second, how many liters of rn, measured at stp, are produced per day by 9.64 g of ra?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3
You know the right answer?
Radon (rn) is the heaviest and the only radioactive member of group 8a(18), the noble gases. it is a...
Questions




History, 26.08.2019 08:30

Mathematics, 26.08.2019 08:30

Biology, 26.08.2019 08:30

Mathematics, 26.08.2019 08:30

History, 26.08.2019 08:30


History, 26.08.2019 08:30

Spanish, 26.08.2019 08:30


Mathematics, 26.08.2019 08:30

Mathematics, 26.08.2019 08:30

History, 26.08.2019 08:30


Mathematics, 26.08.2019 08:30


Mathematics, 26.08.2019 08:30

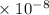 L.
L.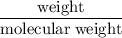
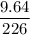
 Avagadro number
Avagadro number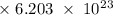
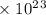
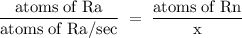
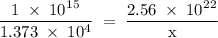
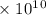 atoms/sec.
atoms/sec.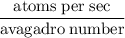
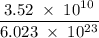 moles
moles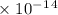 mol/sec.
mol/sec.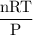
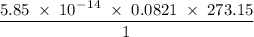
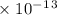 liter/sec.
liter/sec.

