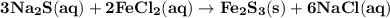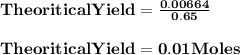Chemistry, 29.07.2019 09:00 josephaciaful
How many milliliters of 0.200 m fecl3 are needed to react with an excess of na2s to produce 1.38 g of fe2s3 if the percent yield for the reaction is 65.0%? 3 na2s(aq) + 2 fecl3(aq) → fe2s3(s) + 6 nacl(aq)?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:00
Surface currents are caused by blank space . question 14 options: surface currents are caused by? differences in water temperature high salinity differences in density wind forces
Answers: 1

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 22.06.2019 15:30
Plz me ! 1 which of earths spheres contains most of its mass? a atmosphere b hydrosphere c geosphere* d biosphere 2 erosion and weathering are examples of which types of forces? a constructive forces b destructive forces* c gravitational forces d inertia-related forces 3 which of the following statements about earths atmosphere is true? a earths atmosphere contains 78% water vapor which is essentail to life b earths atmosphere contains 21% oxygen c earths atmosphere contains carbon dioxide which all life forms require d earths atmosphere allows radiation from the sun to pass through it and warm earths surface* 4 the strenght of the force of gravity between two objects is determined by which of the following factors? select all that apply a the messes of the objects* b the distance between the objects* c the volumes of the objects d the surface area of the objects 5 earth and moon are kept in there respective orbits due to the influence of a inertia b gravity c gravity and inertia* d neither gravity or inertia if you answer all questions right i will give
Answers: 1

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3
You know the right answer?
How many milliliters of 0.200 m fecl3 are needed to react with an excess of na2s to produce 1.38 g o...
Questions




Mathematics, 23.04.2020 18:02

Social Studies, 23.04.2020 18:02


Mathematics, 23.04.2020 18:02

Mathematics, 23.04.2020 18:02

History, 23.04.2020 18:02



Mathematics, 23.04.2020 18:02




History, 23.04.2020 18:02

Mathematics, 23.04.2020 18:02

Mathematics, 23.04.2020 18:02

Social Studies, 23.04.2020 18:02

History, 23.04.2020 18:02

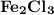 is needed to react with an excess of
is needed to react with an excess of 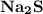 to produce 1.38 g of
to produce 1.38 g of 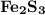 .
.