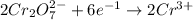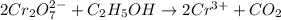Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 08:00
What are the similarities of physical and chemical change ?
Answers: 1

Chemistry, 22.06.2019 10:00
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3
You know the right answer?
Balance the following redox equation, identifying the element oxidized and the element reduced. show...
Questions

English, 09.06.2021 14:00


Physics, 09.06.2021 14:00

World Languages, 09.06.2021 14:00

Chemistry, 09.06.2021 14:00


Social Studies, 09.06.2021 14:00

Mathematics, 09.06.2021 14:00


English, 09.06.2021 14:00


Geography, 09.06.2021 14:00


Mathematics, 09.06.2021 14:00

Business, 09.06.2021 14:00


Mathematics, 09.06.2021 14:00

Social Studies, 09.06.2021 14:00

Physics, 09.06.2021 14:00

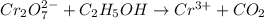
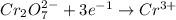
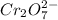 is +6.
is +6.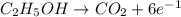
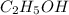 is -2 and in
is -2 and in 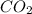 oxidation state of C is +4.
oxidation state of C is +4.