
Chemistry, 28.07.2019 04:00 aleyshamar14p95561
Co(g) + 2 h2 --> ch3oh 2.50 g of hydrogen is reacted with 30.0 l of carbon monoxide at stp. 1. what is the limiting reactant? *hint: only list the element symbol* 2. what mass of ch3oh is produced? *hint: only list the grams* 3. how much excess is left over? *hint: only list the grams*

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Plz mark brainliest 30 points1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s.2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2


Chemistry, 23.06.2019 00:30
Maya wrote if you step to describe how carbon circulates between the atmosphere and living organisms
Answers: 1
You know the right answer?
Co(g) + 2 h2 --> ch3oh 2.50 g of hydrogen is reacted with 30.0 l of carbon monoxide at stp. 1. w...
Questions

Geography, 24.01.2021 22:40


Social Studies, 24.01.2021 22:40

Geography, 24.01.2021 22:40

History, 24.01.2021 22:40







Mathematics, 24.01.2021 22:40

Physics, 24.01.2021 22:40


English, 24.01.2021 22:40





SAT, 24.01.2021 22:40

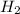
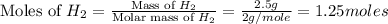
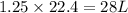 volume of hydrogen gas
volume of hydrogen gas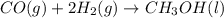
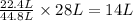 of carbon monoxide gas
of carbon monoxide gas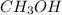 is, 20 grams
is, 20 grams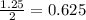 mole of
mole of 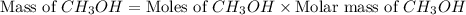
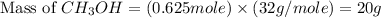
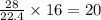 gram of carbon monoxide gas
gram of carbon monoxide gas

