Chemistry, 27.07.2019 09:30 brasherfamily14
What volume of a 1.0 m hcl is required to completely neutralize 25.0 ml of a 1.0m koh

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2

Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3

Chemistry, 23.06.2019 04:00
What changes occur in the reaction indicated by the equation? check all that apply. the hydrogen nucleus loses protons. the oxygen nucleus gains protons. the bond in h2 is broken, and new bonds are formed between hydrogen and oxygen atoms. each electron associated with a hydrogen atom is shared with an oxygen atom.
Answers: 3

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
What volume of a 1.0 m hcl is required to completely neutralize 25.0 ml of a 1.0m koh...
Questions

Mathematics, 23.10.2020 06:01



Biology, 23.10.2020 06:01

Arts, 23.10.2020 06:01

History, 23.10.2020 06:01

Mathematics, 23.10.2020 06:01


Mathematics, 23.10.2020 06:01

Mathematics, 23.10.2020 06:01

Biology, 23.10.2020 06:01


History, 23.10.2020 06:01




Chemistry, 23.10.2020 06:01


Mathematics, 23.10.2020 06:01

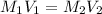
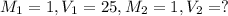
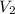 )
)